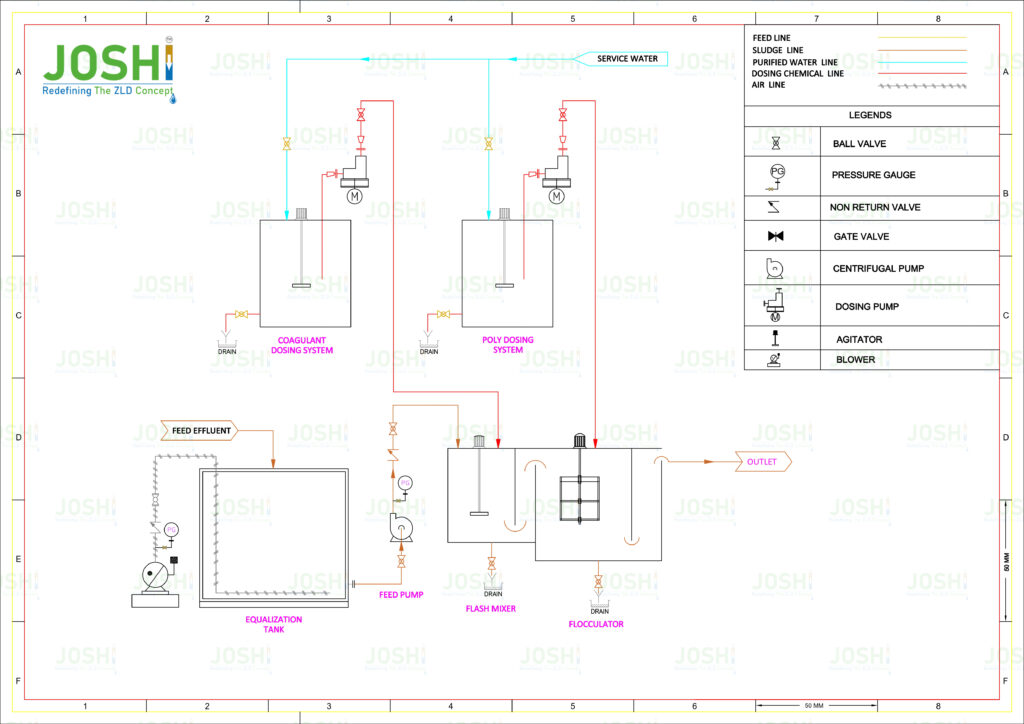
Coagulation - flash mixer tank fitted with a high speed mixer
It is an essential step in the treatment process to facilitate the removal of suspended solids, organic matter, and certain dissolved substances and enhancing the efficiency of subsequent treatment steps, such as sedimentation, filtration, or disinfection.
Process
Coagulation is a chemical process use in wastewater treatment to destabilize and aggregate particles suspend in a liquid, allowing them to form larger and more easily removable flocs. During coagulation, a coagulant, typically a metal salt such as aluminum sulfate (alum) or ferric chloride, is add to the wastewater.
The coagulant’s positive ions neutralize the negative charges on the particles, reducing the electrostatic repulsion between them. As a result, the destabiliz particles come closer together and begin to aggregate.
Coagulation helps in the removal of a variety of impurities, including suspended solids, colloidal particles, turbidity, algae, certain dissolve organic compounds, and heavy metals. It also aids in the removal of pathogens, as coagulation can assist in the agglomeration and subsequent removal of microorganisms.
Factors Affecting Process
The effectiveness of coagulation is influence by factors such as the choice and dosage of coagulant, pH levels, mixing intensity, and the characteristics of the particles or colloids present in the water or wastewater.
Cascade aerators are also use to remove volatile gases, such as carbon dioxide and hydrogen sulfide, from the water. These gases can contribute to unpleasant odors and can be harmful to aquatic life if present in excessive amounts. By exposing the water to air, cascade aerators facilitate the release of these gases, improving water quality.
Targeted Impurities
- Suspended Solids
- Colloidal Particles
- Turbidity
- Algae
- Certain Dissolved Organic Compounds
- Heavy Metals
- Pathogens
- Microorganisms